Consensus splice site11/24/2023 The vast majority of this intergenic transcription is degraded soon after synthesis and this is reflected in the fact that at steady state levels protein coding RNA is present at levels 25–100 fold greater than intergenic RNA. In mammalian cells, intergenic transcription accounts for a large fraction of the total nascent RNA output, approximately equal to the amount of protein-coding RNA. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.Ĭompeting interests: Alexander Palazzo is a PLOS ONE Editorial Board member, and that this fact does not alter the authors' adherence to PLOS ONE Editorial policies and criteria. įunding: This work was supported by a grant from the Canadian Institutes of Health Research to AFP (FRN 102725). Genome sequences are available at Figshare. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are creditedĭata Availability: All relevant data, except for genome sequences, are within the paper and its Supporting Information files. Received: OctoAccepted: FebruPublished: March 31, 2015Ĭopyright: © 2015 Lee et al. PLoS ONE 10(3):Īcademic Editor: Zhi-Ming Zheng, National Institute of Health - National Cancer Institute, UNITED STATES Our data suggests that the presence of the 5’splice site motif in mature RNAs promotes their nuclear retention and may help to distinguish mRNAs from misprocessed transcripts and transcriptional noise.Ĭitation: Lee ES, Akef A, Mahadevan K, Palazzo AF (2015) The Consensus 5' Splice Site Motif Inhibits mRNA Nuclear Export. This motif is also depleted from the beginning and ends of the 3’terminal exons of spliced mRNAs, but less so for lncRNAs. Genome wide analysis of human mRNAs, lncRNA and eRNAs indicates that this motif is depleted from naturally intronless mRNAs and eRNAs, but less so in lncRNAs. The motif, however, does not disrupt splicing or the recruitment of UAP56 or TAP/Nxf1 to the RNA, which are normally required for nuclear export. Our new data indicates that this motif also inhibits nuclear export and promotes the targeting of transcripts to nuclear speckles, foci within the nucleus which have been linked to splicing. Previously it was shown that when this motif is retained in the mRNA, it causes defects in 3’cleavage and polyadenylation and promotes mRNA decay. This element has the same sequence as the consensus 5’splice site motif that is used to define the start of introns. 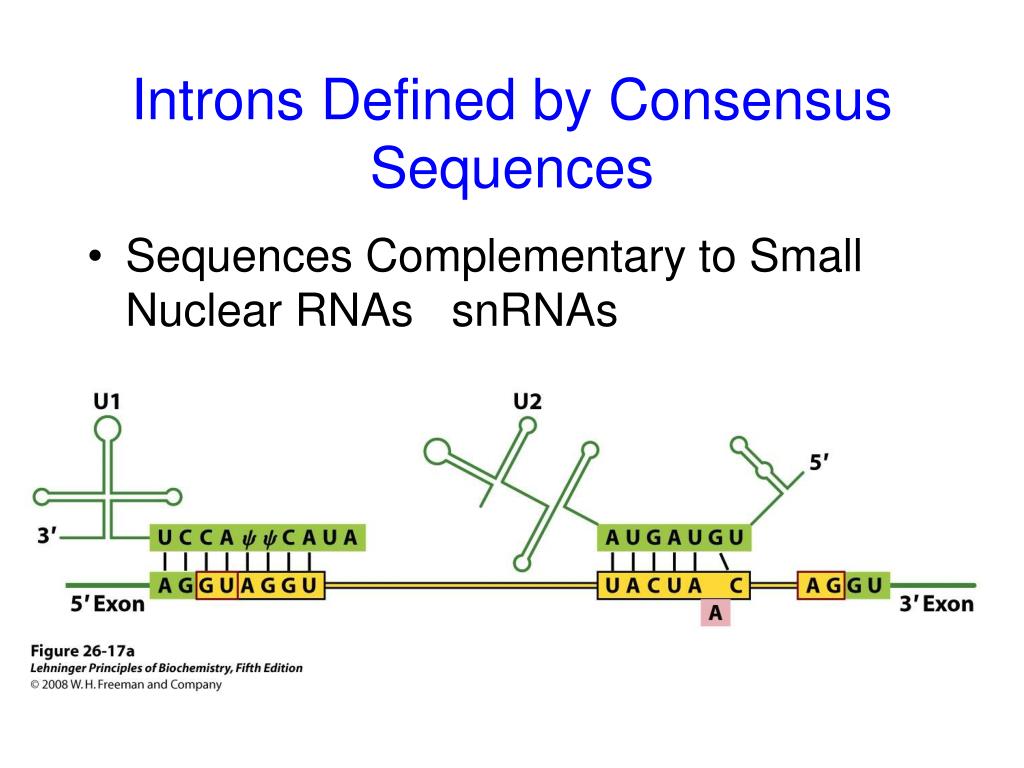
We have mapped an element, which when present in the 3’terminal exon or in an unspliced mRNA, inhibits mRNA nuclear export. In eukaryotes, mRNAs are synthesized in the nucleus and then exported to the cytoplasm where they are translated into proteins.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
